Campaigners have threatened legal action to stop an NHS gender trial which will inject dozens of children as young as ten with puberty blockers.
The trial, which is part of a £10.7 million ‘Pathways’ research programme and will study the effects of the banned puberty suppressing drugs on young people’s brains, was controversially green lit yesterday.
For the first time ever, potential brain damage triggered by the drugs will be studied by monitoring young children’s brains.
They are expected to be put on the drugs for up to two years and monitored until they reach early adulthood. Trial findings could take four years.
Use of the drugs – which delay or prevent puberty from taking place in young people with gender dysphoria – was banned last year after the Cass Review – led by Dr Hilary Cass – ruled that there was not enough clinical evidence to say whether their use was dangerous or not.
Dr Cass, who said she was subject to ‘vile’ online abuse after her findings were published and was advised not to use public transport because of fears over her personal safety, recommended that further research was needed to assess whether the drugs were safe.
Opponents including Maya Forstater of charity Sex Matters, say the treatment could lead to irreversible lifelong harm and it is ‘foolish and unethical’ to expose children to ‘experimental treatment’.
They have threatened a legal challenge to the trial – which is expected to start in the New Year – to stop it from going ahead.

Maya Forstater, CEO of sex-based rights charity Sex Matters, which has threatened a legal challenge to an NHS gender trial which will inject children with puberty blockers
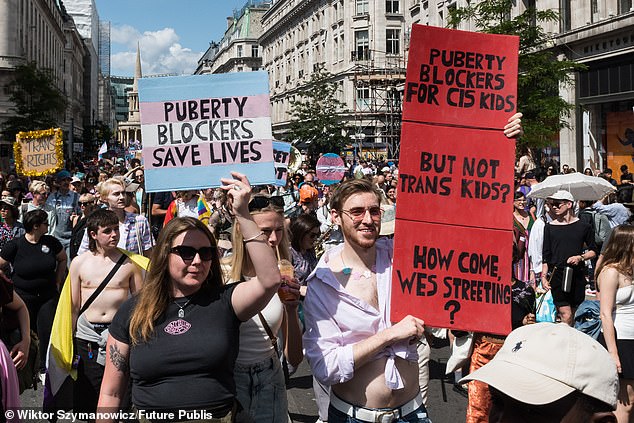
Whether or not children should be allowed puberty blockers has been at the centre of several pro-trans protests in London and other UK cities
Keira Bell, who has previously taken part in legal action against the Tavistock Gender Clinic which gave her puberty blockers as a teenager, said today that she and others will launch a legal challenge ‘unless this trial is immediately halted on the basis that it is unlawful’.
Ms Bell, who later said she should never have been put on the drugs as a teen, claimed on social media: ‘Over 500 children are expected to be put on these drugs which have indicated they cause cognitive impairment amongst other life-altering effects.’
Fellow campaigner James Esses said the drugs caused physical and emotional irreversible damage and ‘putting more children on a path towards such harm’ was like ‘leading lambs to slaughter’.
Meanwhile campaign group Transgender Trend said there ‘was no justification to conduct this experiment again’.
In response to the growing criticism, Health Secretary Wes Streeting waded in to defend the trial this morning.
He said: ‘Children with gender incongruence deserve safe, compassionate and effective care. That healthcare must always be led by evidence.’
Professor Emily Simonoff, Chief Investigator of the study said she recognised views were polarised on the topic but ‘right in the middle there are young people and their parents who are attending services, who have gender incongruence and don’t know what to do’.
Ms Simonoff, Professor of Child and Adolescent Psychiatry at King’s College Hospital’s which will undertake the trial using 226 boys and girls under 16, around half of whom are expected to be injected with the drugs, said: ‘This study alongside the other Pathways studies is really aiming to provide the same level of evidence for young people with gender incongruence as we provide for any other disorder for people attending NHS services.’

Dr Hilary Cass said in her Cass Review that the quality of previous studies claiming puberty blockers have beneficial effects was ‘poor’

Professor Emily Simonoff, who is running the NHS gender trial which will inject dozens of children with puberty blockers, says she is ‘very proud’ to be in charge of the landmark study
She told the BBC today that the trial would be subject to the same strict approvals and regulations as any other medical trial in the UK, commenting:
‘I am very proud to be the chief investigator for the Pathways study. I recognise it’s an extremely important question and it’s very important for the young people and their parents who at the moment are really uncertain what kind of healthcare to opt for.
‘What we hope is that the trial plus the wider studies will allow us to give young people experiencing gender incongruence and their families and those who are treating them much better information about treatment moving forwards.’
It is understood that children selected for the trial will be under 16s currently accessing gender services who have undergone intensive medical or psychological screening.
And Professor Simonoff said that as well as looking ‘at the balance between possible benefits for mental health and quality of life and the possible risks and harm to physical health’ of the drugs ‘one of the really important things we are doing is looking at whether there are any possible risks to brain development by doing the first ever study looking at cognitive development – that’s thinking, learning and memory’.
She said: ‘We will be scanning the brains of young people who are getting puberty suppressing hormones and those who are not – that’s the first time that’s ever been done.’
Professor Simonoff said the size of the trial was necessary to ensure ‘the sample is large enough that we don’t miss true effect if it’s present and at the same that it is large enough that we don’t conclude a positive or beneficial effect that isn’t present’.
She said: ‘This study has gone through all the usual approvals and regulatory scrutiny which involves the Human Research Authority and the Medicines and Healthcare Regulatory Agency (MHRA).
‘The UK has very high levels of scrutiny of clinical trials to make sure that they are properly conducted but also we are looking at young people’s psychological state, emotional wellbeing, physical health and quality of life as well as any possible adverse effects here. We are doing that in a very structured and standard way.’
She told Radio 4’s Today programme that the study would be advised by ‘people with lived experience – both people with trans experience and also parents and caregivers who come from a diversity of experiences of gender diversity or gender incongruence’.
‘We are not expecting a one size fits all finding,’ she said.
Recruitment for the trial will be the only way for under 18s in the UK to get puberty blockers after the closure of the controversial Tavistock Gender Identity Development Service (GIDS) in London last year. The clinic had been giving puberty blocking drugs to young children












