Two of the UK’s leading transplant centres put patients at risk by using a heart device feared to be linked to higher death rates than a rival product, it has emerged.
The NHS raised concerns about the device in 2018, with half who were fitted with the product dying within three years.
Despite this, Newcastle-based Freeman Hospital and London‘s Harefield continued to use the pump.
Its manufacturer, Medtronic, withdrew the product in 2021 on safety grounds. It also cited a malfunction where pumps would delay or fail to restart after they were stopped.
Both hospitals were aware their leading cardiologists were paid consultants for the device’s manufacturer, the BBC reported.
A preliminary audit by the NHS in 2019 revealed that of the 119 patients who had received the Medtronic device, 54 had died within two years.
In contrast, 15 out of 97 patients who received the Abbott pump had died over the same period.
The device, which can be offered to patients who may be waiting for a transplant or are ineligible for one, helps the heart pump around the body.
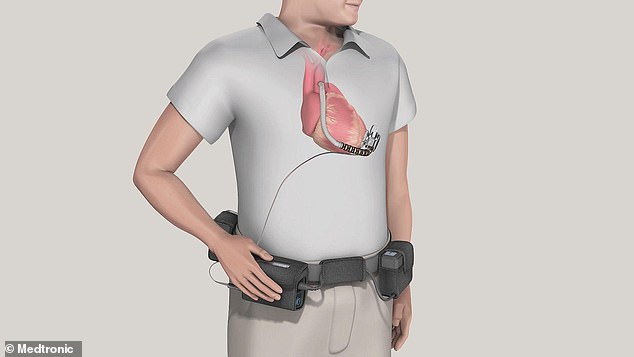
The device, which can be offered to patients who may be waiting for a transplant or are ineligible for one, helps the heart pump around the body. The NHS raised concerns about the device in 2018, with half who were fitted with the product dying within three years
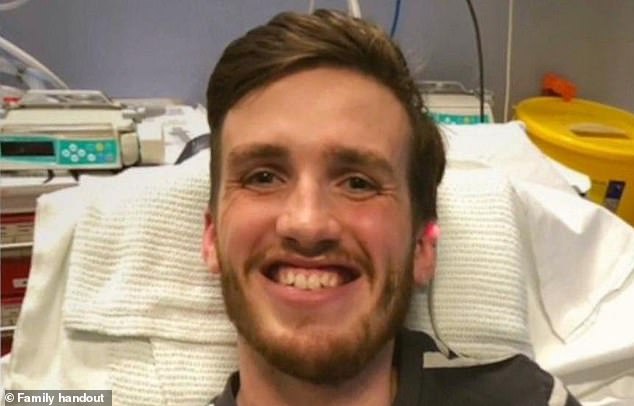
Greg Marshall, a fit and healthy man who had ambitions of joining the Royal Marines, was one of the patients fitted with the Medtronic device in 2019. However, in September 2023, he unexpectedly went into cardiac arrest and died aged 26
Greg Marshall, a fit and healthy man who had ambitions of joining the Royal Marines, was one of the patients fitted with the Medtronic device in 2019 after he suffered an acute heart failure.
He was offered the Medtronic device by the Freeman Hospital, several months after the NHS data set was available.
Greg agreed to the surgery but suffered a stroke due to a significant complication. He suffered a stroke and lost movement down the left-hand side of his body, and his speech was significantly impaired.
During his slow recovery heart device suddenly stopped working in July 2020 and it failed to switch back on when he tried to restart it.
He was rushed to the hospital, but the device could not be fixed, and it remained inside him as he was refused another surgery, terrified of having another stroke.
His heart continued to function, and he was placed on a transplant waiting list.
However, in September 2023, he unexpectedly went into cardiac arrest and died aged 26.
‘It was a massive shock to us all,’ his mother, Tessa Marshall, said.
She said they were not presented with the long-term risks and that an existing patient was even brought in to tell them of its benefits.
‘I kick myself now, for not doing any more research,’ Ms Marshall said.
The family say the health professional responsible for Greg’s care, Prof Stephan Schueler, did not declare to them his financial relationship with Medtronic, even though it is required by the doctor’s regulator, the General Medical Council (GMC).
Prof Schueler said: ‘There was never a financial incentive nor any salary arrangements with Medtronic for me or anybody else in our team to choose one device over the other.’
Another transplant centre, the Royal Papworth Hospital in Cambridge, voluntarily chose to stop using the Medtronic device in February 2018 after it opted to use an alternative pump following the results of two controlled trials.
However, Harefield Hospital continued to solely use the Medtronic device until early 2021.
The Freeman Hospital continued until June 2021, when it was withdrawn from sale ‘in the interest of patient safety’.
The regulator, Medicines and Healthcare products Regulatory Agency, continued to approve the device for use, but it had not been informed by the NHS of the 2019 data.
Figures released under the Freedom of Information Act revealed that between October 2018 and June 2021, the mortality rate of the Medtronic device was two-and-a-half times higher than the Abbott device.
Forty-nine per cent of those who were given the Medtronic device in that time died within three years, while 19 per cent of recipients of the Abbott device died in the same timeframe.
Both hospitals said their decisions to continue using the pump were based on ‘complex clinical decisions’, adding there was ‘no clear grounds’ at the time to believe that Abbott’s device was inferior to Medtronic.